Columnist Justin Freid explains the basics of getting your PPC campaigns up and running in the highly regulated industry of prescription drug advertising.

One out of every 20 searches on Google is health-related. That means that many of us are heading to Dr. Google before heading to see our primary care physician. What the searchers do not see is the complex set of rules, regulations and processes that many pharmaceutical companies must go through to leverage paid search.
Though most are familiar with the FDA’s role in deciding which medications are approved for use, many are not aware that it also oversees advertising for prescription drugs.
The United States is one of the few countries that allow direct-to-consumer advertising for pharmaceutical drugs, and the FDA plays a key role in making sure any type of advertising by pharmaceutical brands is accurate and not deceptive. It also ensures that any claims made are backed up with information about the potential side effects or risks.
For an SEM campaign to be launched, a strict set of rules must be followed. Many of the rules come from the FDA’s guidance on Reminder Ads, while others are set by legal and regulatory teams within the specific pharmaceutical companies.
FDA guidance overview
The FDA has extensive advertising guidance for prescription drugs, some of which is applicable to SEM. The issue is, some points within the guidance are interpreted differently by individual pharma companies because guidelines are general enough to apply to multiple forms of advertising (display creative, SEM creative).
That means that, as new and innovative forms of advertising are created, the guidance from the FDA is often applied to these new opportunities, even if it does not fit exactly. (Editor’s Note: the legal information provided in this column is just that — legal information, and not legal advice.)
Following are some FDA guidelines and how they apply to SEM.
Fair balance
FDA regulations require that pharmaceutical ads provide a “fair balance” of information on both a drug’s risks and its benefits. According to the FDA website, “This means that the content and presentation of a drug’s most important risks must be reasonably similar to the content and presentation of its benefits.”
This is why, at the end of TV commercials for prescription drugs, you often hear that long list of side effects and risks. This is meant to counterbalance the information given about the drug’s effectiveness in treating a condition — because an ad cannot place greater emphasis on benefits than it does on risks.
Reminder ads
The FDA’s definition of reminder ads is as follows:
Reminder ads give the drug’s name but not the drug’s use. The assumption behind reminder ads is that the audience knows what the drug is for and does not need to be told. A reminder ad does not contain risk information about the drug because the ad does not discuss the condition treated or how well the drug works.
In other words, reminder ads avoid the need for “fair balance” of information because they contain neither benefits nor risks — just the name of the drug, which serves as a “reminder” for consumers who are already familiar with it.
These ads cannot explain — or even suggest — what the drug does, what disease it treats, what the recommended dosage is, or how well the drug works; however, they do not have to include any risk information about the drug, either. These ads must mention the drug’s brand name (if applicable) and its generic name.
Because of character limits in search text ads, this guidance is often applied. Here is an example of what a reminder ad would look like:

As mentioned above, this ad falls under the reminder ad guidance because it contains the drug’s brand name but does not address the disease or condition it treats. If it did, “fair balance” would apply, and the risks would need to be disclosed. This obviously is not possible, as it would be significantly longer than standard text ad character limits.
It is important to note that the reminder ad guidance is not directed solely at SEM efforts and is applicable to many forms of advertising.
Space limitations
The FDA has also published guidance regarding “Internet/Social Media Platforms With Character Space Limitations.” This guidance can also potentially be applied to search text ads and in some ways contradicts the reminder ad policy.
This guidance restates that if a brand chooses to disclose a benefit, then the risk information must follow. However, it also explains that if there are not enough characters to allow the full set of risk information, then the risks must be listed in order of seriousness.
Here is the specific section addressing this:
At a minimum, a firm should communicate the most serious risks associated with the product together with the benefit information within the individual character-space-limited communication.
Throughout the document, the FDA states that if the pharmaceutical company feels they cannot accurately present risk information in the allotted space, they should reconsider showing their brand name and benefit information in that specific ad unit.
When this guidance is applied to a text ad, it could potentially resemble the following:
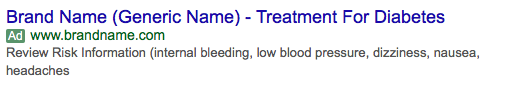
Many pharmaceutical companies are not comfortable leveraging this within paid search efforts and do not use this format. Almost all companies ensure text ads stay within the reminder ad guidance mentioned above.
Paid search for ‘black box’ drugs
A boxed warning appears on a prescription drug label for any drug that has a serious risk associated with it. If a drug happens to have a side effect that could cause death, addiction or serious injury, it is often labeled as “black box.”
(Note: Advertisers are not allowed to use reminder ads for “black box” drugs.)
For many years, Google and Bing both had specific ad formats for black box drugs. This included an additional line of text, similar to current call-out extensions, that said: “Click to see full safety and prescribing information, including boxed warning. More info.”
The “more info” part of the ad would link to the important safety information (ISI) page on the brand website. This ad format was not accessible within the AdWords or Bing Ads interface and needed to be set up by Google or Bing reps.
This ad format was done away with in mid-2015. Now many brands with boxed warnings utilize similar text within their description lines and drive users to a page that contains ISI information.
An example of what a black box ad may look like is as follows:

Changes to Vanity URL usage for pharma advertisers
As mentioned above, the FDA requires you to list risk information if you call out the disease or condition the drug treats within a text ad. This caused the pharmaceutical industry some problems with trying to make a text ad relevant to a search query.
If someone searched for heart failure medication and you wanted to include the term “heart failure medication” within your ad, you wouldn’t be able to unless you either disclosed the risks or removed the drug’s brand name. With both Google and Bing showing the destination URL within ads — which was often branded — this caused a big issue for brand ads.
To circumvent this issue, both Google and Bing previously allowed pharma brands to utilize Vanity URLs. These essentially were unbranded URLs that redirected to a brand website. For example, www.heartfailuremeds.com would redirect to www.brand.com. This way, the ad would not show the brand name but would show www.heartfailuremeds.com instead. This practice, of course, is against regulations for all advertisers outside of pharma.
In early 2016, Google did away with this option, as they called out the practice as deceptive to searchers. Now, advertisers can utilize a select set of “display URL options.”
Currently, pharma advertisers can choose from the following within AdWords:

This was a hot topic within the SEM community in 2016, as many PPC experts were surprised Google was allowing advertisers to execute something that is not available to any other industry.
The legal review process
Every pharmaceutical company has an internal legal and regulatory team responsible for reviewing myriad materials. From websites and in-office pamphlets to videos and SEM ads, all marketing materials go through a rigorous review process.
One potential issue agencies run into is how different companies, and even different reviewers within the same company, interpret FDA guidance. A process or approach to text ads that works for one client may not be acceptable for another.
As a best practice, working alongside legal and regulatory teams to ensure you understand their internal processes while also helping educate them on the specific channel you manage can help make the process as smooth as possible.
Working with the engines
Getting a new pharma SEM campaign off the ground is tough enough when you have to jump through the legal and regulatory hoops, but there are extra steps that must be taken with the search engines as well.
Unfortunately, you cannot just set up a new campaign and get it up and running right away. When flagged as a pharmaceutical brand, the website that traffic is driving to must be whitelisted internally at the engines to qualify for a campaign to go live. Without this, ads will be disapproved immediately.
Thankfully, both Bing and Google have dedicated teams that fully understand the pharmaceutical rules and regulations. Similar to other industries, spend is a factor of receiving dedicated support. In some certain cases, smaller startup pharmaceutical companies with low or no historic spend will have to utilize the AdWords Hotline to get their campaigns up and running.
[Read the full article on Search Engine Land.]
Some opinions expressed in this article may be those of a guest author and not necessarily Marketing Land. Staff authors are listed here.
Marketing Land – Internet Marketing News, Strategies & Tips
(118)